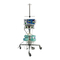
Indications For Use; Intended Users; Contraindications - Hologic Fluent Pro Bedienungsanleitung
Verfahrens-kit
Inhaltsverzeichnis
Fluent Pro Procedure Kit Components
4
In-FloPak
5
Out-FloPak
6
Tissue Trap
I
U
NDICATIONS FOR
SE
The Fluent Pro fluid management system is
intended to provide liquid distension of the uterus
during diagnostic and operative hysteroscopy, and
to monitor the volume differential between the
irrigation liquid flowing into and out of the uterus
while providing drive, control, and suction for
hysteroscopic morcellators.
The Fluent Pro Procedure Kit should only be used
with the Fluent Pro fluid management system.
I
U
NTENDED
SERS
The gynecologist should be trained in diagnostic and
therapeutic hysteroscopy, resection, and removal of
gynecological tissue.
I
NTENDED USE ENVIRONMENT
The Fluent Pro fluid management system is
designed to be used in operating rooms, ambulatory
surgical
centers,
environments.
P
T
G
ATIENT
ARGET
ROUP
The intended patient population for the Fluent Pro
fluid management system is dependent on the
Indications for Use and Contraindications; it is not
The In-FloPak pulls clean fluid from the Fluid Bag. It contains the Fluid Bag Tube
and the hysteroscope Inflow Tube. It fits securely into the blue Fluent Pro In-
FloPak receptacle on the front left side of the fluid management system. These
connections allow the transfer of distension fluid from a Fluid Bag to the
hysteroscope inflow channel. The flow of fluid is monitored and controlled using
the Touchscreen to maintain the pressure at a specified setting.
The Out-FloPak drains waste fluid from the hysteroscope Outflow Channel, the
MyoSure
®
Tissue Removal Device (TRD) tube, and the Under-Buttocks Drape
(UBD) Tube into the Waste Bag. It fits securely into the yellow Fluent Pro Out-
FloPak receptacle on the front right side of the fluid management system. These
connections allow the transfer of fluid from the hysteroscope Outflow Channel,
the MyoSure Tissue Removal Device (TRD), and the Under-Buttocks Drape
(UBD) Port to the Waste Bag.
The Tissue Trap is designed to capture resected tissue throughout the
procedure to allow the tissue to be sent to pathology. The Tissue Trap Canister
contains the Tissue Trap that captures the resected tissue. Please monitor the
Tissue Trap so it does not overfill.
and
physician's
limited by age, weight, or other health conditions
unrelated to hysteroscopic contraindications.
C
ONTRAINDICATIONS
The system may not be used to introduce fluids into
the uterus when hysteroscopy is contraindicated.
See the operator's manual of your hysteroscope for
absolute and relative contraindications.
The Fluent Pro fluid management system should not
be used to remove pathologies from pregnant
patients or patients exhibiting pelvic infection,
cervical malignancies, or previously diagnosed
endometrial cancer.
WARNING!
Fluent
disposables. Do not re-use or reprocess any of the
components contained within the single-use Fluent
Pro Procedure Kit. Re-use or reprocessing of such
components may result in infection or product failure
due to material degradation.
office
WARNING!
For your own safety and that of your patient, use only
Fluent Pro accessories.
See the Fluent Pro System IFU for additional
warnings and precautions.
Pro
Procedure
Kits
are
single
use
2
Inhaltsverzeichnis