Coltene ACCU-TRAC Gebrauchsanweisung
Precision die system
Inhaltsverzeichnis
Verfügbare Sprachen
Verfügbare Sprachen
Instructions for Use
Intended Purpose
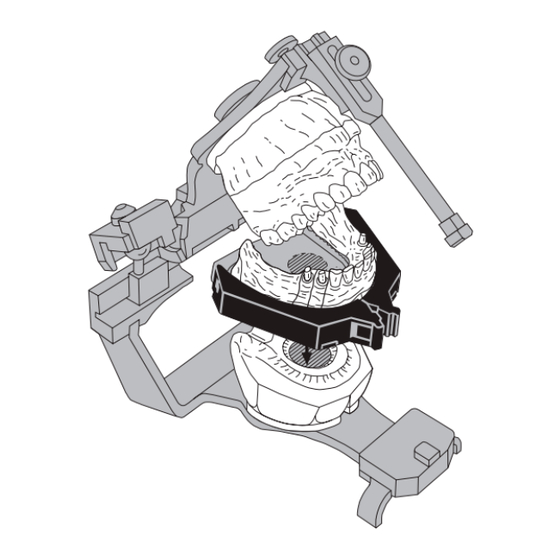
ACCU-TRAC Precision Die System is intended to act as a
base and retention system for models and dies for use in the
creation of dental restorations. These models and dies can be
used as tools for planning for the appropriate restoration such
as crowns, bridges, implants, combinations, or other dental
prostheses.
Intended User
The intended user population are dentists, hygienists, and
laboratory technicians in a dentist office. The ACCU-TRAC
Precision Die System is not intended to be used by the general
population.
Intended Patient Population
The ACCU-TRAC Precision Die System is not used on patients
or by patients. It is used by dentists, hygienists, and laboratory
technicians in a dental office.
Clinical Benefits
The ACCU-TRAC System provides the clinical benefit of effective
fabrication of dental models and dies for the creation of dental
restorations.
Contraindications
The ACCU-TRAC System does not have any contra-indications.
Residual Risks
Failure to follow instructions provided in this IFU may result in
inaccurate or incomplete model fabrication.
Avoid using excess model material to ensure proper function of
the magnetic retention system.
Use caution when operating locking arms to avoid breakage,
damage to the model, pinching, or other hand injury.
TYPE
Standard Packaging
Economy Packaging
Accessories
Caution: Federal law restricts this device to sale by or on
the order of a licensed healthcare practitioner.
ACCU-TRAC®
Precision Die System
ORDERING INFORMATION
ITEM
Introductory Kit
Refill Kit
Introductory Kit
Refill Kit
Articulation Keepers
Locking Arms
Model Ejectors
Articulation Spacers
Accu-Trans
®
Transport System
Avoid using this device around magnets as this may interfere with
the magnetic retention system and may damage the model.
Avoid using this device around electronic devices as the magnets
may cause electromagnetic interference.
Symbols Glossary
Symbol
Meaning
Consult the instructions for use
Medical Device Indicator: this product meets the
definition of a medical device
Sale Restricted: the purchase of the device is
restricted to healthcare practitioners only
Date of manufacture
COLTENE catalog number
CE Mark
Indicates the entity importing the medical device
The entity that manufacters the medical device
Batch code
Authorized Representative in the European
Community
Notice: Report any serious incident that has occurred in relation to
this device to the manufacturer and the competent authority of the
Member State in which the user and/or patient is established.
Upon request, a hardcopy of the IFU will be supplied free of
charge.
CAT NO.
DESCRIPTION
AT6
6 Accu-Trac Precision Die Trays
6 Pouring/Ejector Bases
25 Articulation Keepers
25 Return Request Cards
25 Patient Identification Labels
ATR12
12 Accu-Trac Precision Die Trays
50 Articulation Keepers
50 Return Request Cards
50 Patient Identification Labels
AT72
12 AT6 Kits, 72 Units
ATR144
12 ATR12 Kits, 144 Units
ATK300
300 Articulation Keepers
300 Return Request Cards
300 Patient Identification Labels
ATK25
25 Articulation Keepers
ATA20
20 Sets Locking Arms
ATE12
12 Pouring/Ejector Bases
ATS20
20 Articulation Spacers
ATN50
50 Disposable Carrying Cases
50 Plastic Retainers
50 Doctor/Patient Labels
Inhaltsverzeichnis

Inhaltszusammenfassung für Coltene ACCU-TRAC
- Seite 3 Gebrauchsanweisung Verwendungszweck Vermeiden Sie die Verwendung dieses Geräts in der Nähe von Das ACCU-TRAC Precision Die System dient als Basis und Halte- Magneten, da dies das magnetische Haltesystem beeinträchtigen system für Modelle und Stümpfe zur Herstellung von Zahnres- und das Modell beschädigen kann.
- Seite 4 Bereich und schneiden Sie die Stümpfe in paralleler Ausrichtung zu den Schienen. 11. Waschen Sie die Stümpfe sauber und trocknen Sie sie sorgfältig. 12. Setzen Sie die Stümpfe in numerischer Reihenfolge wieder in Accu-Trac ein Abb. 2 und sichern Sie das Sägemodell mit den Verriegelungsarmen.



